Session:: ePosters-Sun800-M4: Colon & Colorectal Cancer
EP278 - Efficacy And Safety of Incorporating Immunotherapy Into Neoadjuvant Chemoradiotherapy (iNCRT) Versus Neoadjuvant Chemoradiotherapy (NCRT) In Locally Advanced Rectal Cancer
Sunday, May 10, 2026
8:49 AM - 8:54 AM ET

Xiaokang Lei
Attending physician
Peking University Cancer Hospital and Institute
Peking University Cancer Hospital
Beijing, Beijing, China (People's Republic)Disclosure information not submitted.
ePoster Presenter(s)
Purpose/Background: Whether iNCRT (including iTNT) can improve the efficacy of LARC remains unclear. The purpose of this study is to evaluate the efficacy and safety of incorporating immunotherapy into neoadjuvant chemoradiotherapy (iNCRT), including incorporating immunotherapy into total neoadjuvant therapy (iTNT), in locally advanced rectal cancer (LARC).
Methods/Interventions: PubMed, Web of Science, EMBASE, and Cochrane Center Register of Controlled Trials were searched from inception to July 18th, 2025. Only randomized controlled trials (RCT) have been conducted. Data were analyzed using a fixed-effects or random-effects model. Primary outcomes included pCR and severe adverse events (SAEs). Additionally, we conducted subgroup analyses and sensitivity analyses to test the stability of the results.
Results/Outcomes: A total of 9 studies with 1150 patients, of whom 611 patients received inCRT, were enrolled for inCRT vs nCRT. And 7 studies including 899 patients, of whom 447 patients received iTNT, were selected for iTNT vs TNT. The pooled rates of pCR were 37.68% (range, 22.00%-59.18%) in the inCRT group and 23.67% (range, 14.04%-32.65%) in the nCRT group. InCRT was associated with a significant higher rate of achieving a PCR with mild heterogeneity (OR=2.06, 95% CI [1.55, 2.72], I2=40%, P< 0.00001). The benefit of iTNT persisted when compared with TNT (41.32% [range, 31.34%-59.18%] vs 24.61% [range, 15.25%-32.65%]; OR=2.16, 95% CI [1.58, 2.96], I2=23%, P< 0.00001). And inCRT was associated with significant improving R0 resection rate compared with nCRT in LARC (95.99% vs 92.53%; P=0.01), other than iTNT vs TNT (96.92% vs 96.02%; P=0.58). No statistically significant differences in rates of OP (4.48% vs 4.83%, OR=0.99, 95%CI [0.53-1.85], I2=39%, P=0.97), rates of SAEs (25.4 % vs 24.5 %; OR 1.25, 95 % CI 0.94-1.67; I² = 0 %; P = 0.13), postoperative complications (35.67% vs 32.75%, P=0.36), and severe postoperative complications (7.32% vs 5.68%; P=0.37s) between participants of inCRT and nCRT were observed. Only NRG-GI002 study reported data on survival, and it showed iTNT was associated with a significant improvement in 3year OS (HR=0.35, 95% CI [0.12, 1.00], P= 0.04), but not DFS (HR=0.95, 95% CI [0.58, 1.55], P= 0.82).
Conclusion/Discussion: The findings of this systematic review and meta-analysis suggest that inCRT or iTNT is a promising alternative strategy in LARC, with improving rates of pCR, MPR, CR and without increasing the incidence of postoperative complications, severe postoperative complications, SAEs compared with standard therapy. However, the immunotherapy-related SAEs should be given importance and be concerned. The long-term outcomes on DFS and OS needs to be furtherly explored.
Efficacy and safety of incorporating immunotherapy into neoadjuvant chemoradiotherapy (iNCRT) versus neoadjuvant chemoradiotherapy (NCRT) in locally advanced rectal cancer
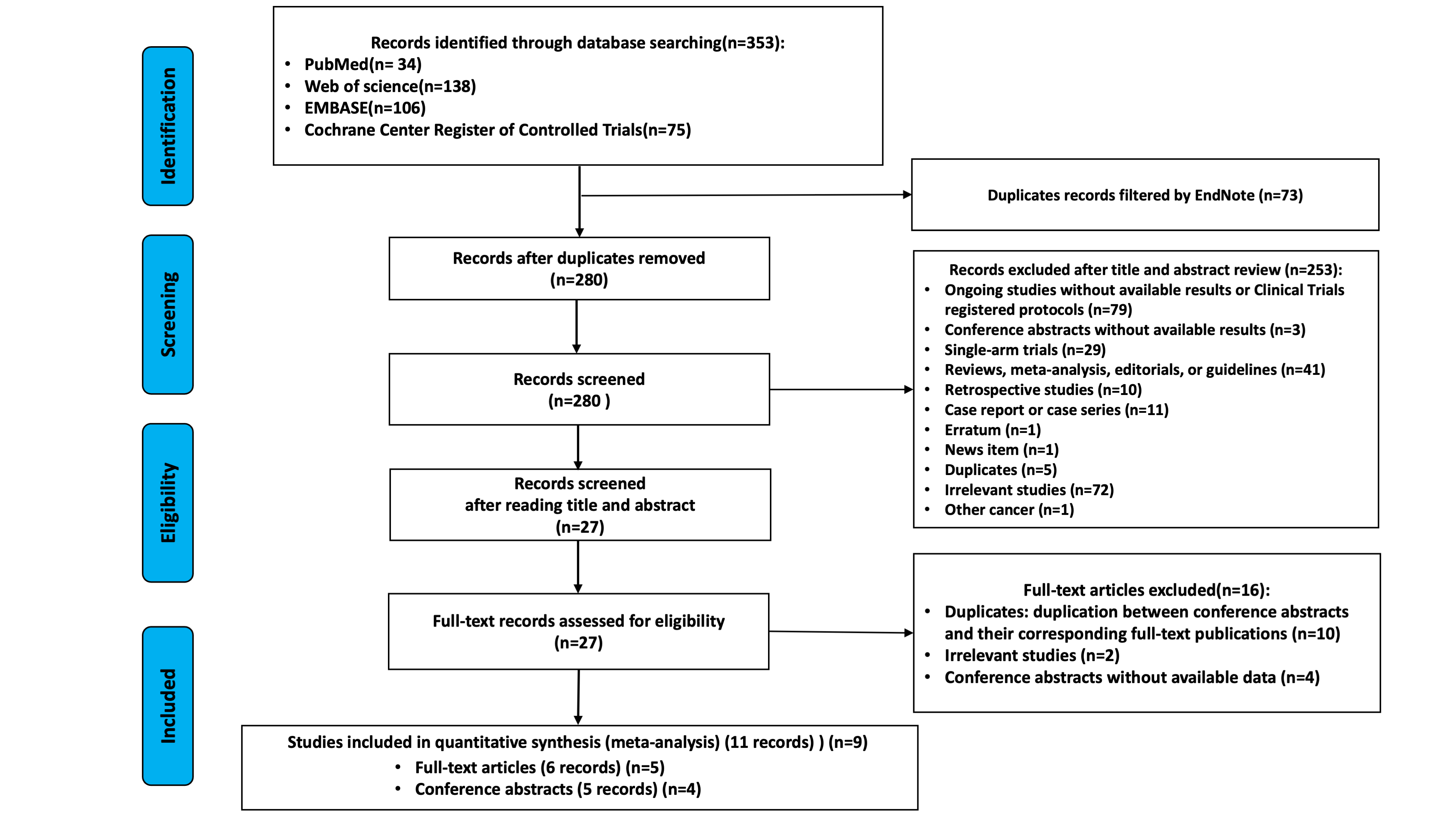
Methods/Interventions: PubMed, Web of Science, EMBASE, and Cochrane Center Register of Controlled Trials were searched from inception to July 18th, 2025. Only randomized controlled trials (RCT) have been conducted. Data were analyzed using a fixed-effects or random-effects model. Primary outcomes included pCR and severe adverse events (SAEs). Additionally, we conducted subgroup analyses and sensitivity analyses to test the stability of the results.
Results/Outcomes: A total of 9 studies with 1150 patients, of whom 611 patients received inCRT, were enrolled for inCRT vs nCRT. And 7 studies including 899 patients, of whom 447 patients received iTNT, were selected for iTNT vs TNT. The pooled rates of pCR were 37.68% (range, 22.00%-59.18%) in the inCRT group and 23.67% (range, 14.04%-32.65%) in the nCRT group. InCRT was associated with a significant higher rate of achieving a PCR with mild heterogeneity (OR=2.06, 95% CI [1.55, 2.72], I2=40%, P< 0.00001). The benefit of iTNT persisted when compared with TNT (41.32% [range, 31.34%-59.18%] vs 24.61% [range, 15.25%-32.65%]; OR=2.16, 95% CI [1.58, 2.96], I2=23%, P< 0.00001). And inCRT was associated with significant improving R0 resection rate compared with nCRT in LARC (95.99% vs 92.53%; P=0.01), other than iTNT vs TNT (96.92% vs 96.02%; P=0.58). No statistically significant differences in rates of OP (4.48% vs 4.83%, OR=0.99, 95%CI [0.53-1.85], I2=39%, P=0.97), rates of SAEs (25.4 % vs 24.5 %; OR 1.25, 95 % CI 0.94-1.67; I² = 0 %; P = 0.13), postoperative complications (35.67% vs 32.75%, P=0.36), and severe postoperative complications (7.32% vs 5.68%; P=0.37s) between participants of inCRT and nCRT were observed. Only NRG-GI002 study reported data on survival, and it showed iTNT was associated with a significant improvement in 3year OS (HR=0.35, 95% CI [0.12, 1.00], P= 0.04), but not DFS (HR=0.95, 95% CI [0.58, 1.55], P= 0.82).
Conclusion/Discussion: The findings of this systematic review and meta-analysis suggest that inCRT or iTNT is a promising alternative strategy in LARC, with improving rates of pCR, MPR, CR and without increasing the incidence of postoperative complications, severe postoperative complications, SAEs compared with standard therapy. However, the immunotherapy-related SAEs should be given importance and be concerned. The long-term outcomes on DFS and OS needs to be furtherly explored.
Efficacy and safety of incorporating immunotherapy into neoadjuvant chemoradiotherapy (iNCRT) versus neoadjuvant chemoradiotherapy (NCRT) in locally advanced rectal cancer

